Primary research - types of study design
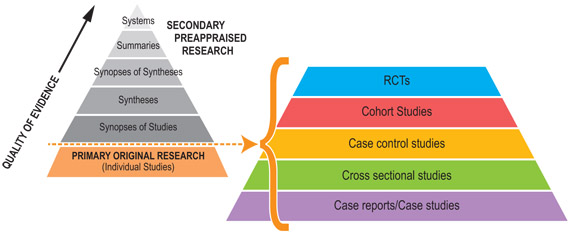
Randomised controlled trial
In a randomised controlled trial (RCT), participants are randomly allocated to different groups – the intervention (such as a drug) group or another group (such as placebo treatment or a different drug). Both groups are followed up for a specified period and analysed in terms of outcomes defined at the outset (death, heart attack, serum cholesterol level, etc.). The studies include methodologies that reduce the potential for bias and allow for comparison between both groups.
RCTs should be used to answer questions such as the following:
- Is this drug better than placebo or a different drug for a particular disease?
- Is a new surgical procedure better than the current practice?
Cohort studies
Two (or more) groups (cohorts) of people are selected on the basis of differences in their exposure to a particular agent or disease (such as a vaccine, a drug, or an environmental toxin), and followed up to see how many in each group develop a particular disease or other outcome. The follow up period is often over several years. These are also known as prospective studies. The ‘outcome’ of interest isn’t apparent at the start of the study.
A cohort study can address clinical questions such as:
- Does smoking cause lung cancer?
- Does oral contraceptive use have an effect on bone mineral density?
Case control studies
Patients who already have a specific condition are compared with people who do not have the condition. They often rely on medical records and patient recall of past exposure for data collection. Case control studies cannot show cause and effect. Showing a statistical relationship between two factors does not mean that one factor caused the other. They are also known as retrospective studies.
A case control study can address clinical questions such as:
- Does the MMR vaccine cause autism?
- Does prolonged use of mobile phones cause brain tumours?
Cross sectional studies
Data are collected at a single time but may refer retrospectively to experiences in the past. A sample of patients is interviewed, examined, or medical records studied to gain answers to a specific clinical question. The exposure and the outcome are determined at the same time.
A cross sectional study can address clinical questions such as:
- What are general practitioners' attitudes to drug information provided by drug companies?
- Is there an association between depression and cigarette smoking?
Case reports and case studies
A case report describes the medical history of a single patient. Case reports are often run together to form a case series, in which the medical histories of more than one patient with a particular condition are described to illustrate an aspect of the condition.
There is no control group with which to compare outcomes.
- Case reports or series may be the best available information on very rare conditions or adverse drug effects
- Information on new or novel treatments which have not yet be studied in an RCT may be first published as case reports or series
Read more about RCTs, cohort studies, and case control in a CIAP online book. [24]
